Transdermal Patch Manufacturing
Diego Lopez – Production Supervisor
The production of transdermal patches is a continuous and artisanal process that consists of 3 stages that reflect how delicate and specific is the production of this niche pharmaceutical form. Let´s review it!
Transdermal patches of the drug-in-adhesive type are pharmaceutical forms that deliver an active ingredient through the transdermal route. They consist of: a support layer, an adhesive matrix containing the active ingredient and a release layer that is discarded when the product is applied to the skin.
The manufacturing process consists of the following stages:
- Mixing
- Coating, Drying and Laminating
- Die-cutting and Packaging
Mixing
It represents the beginning of the manufacturing process and its objective is to achieve the integration of the active and non-active ingredients that make up the formulation.
The first step is to fraction and weight each of the materials that will be part of the mixture, according to what is established in the production methodology.
The following materials are normally involved in this process: API (Active Pharmaceutical Ingredient) and functional and non-functional excipients. The semi-finished product resulting from this phase being the Mix according to the formulation.
At this stage it is important to control the correct dissolution and dispersion of the components to guarantee the homogeneity of the adhesive mixture necessary to ensure the correct dosage and that all the units produced are effective and safe because of the controlled release of the active ingredient.
Coating, Drying and Laminating
Once the mixture is obtained, the following process consists of three sub-stages: first is the coating, which is a continuous process where the mixture is uniformly dispensed on the release liner.
Subsequently, both components go through various drying stations in order to evaporate the volatile components of the mixture, in order to obtain the “adhesive mixture”.
Finally, both components are joined at the exit of the drying stations with the backing film, thus obtaining the multilayer system that forms the transdermal system (backing film-adhesive matrix-release liner).
Typically, the raw materials for this phase are: the mixture (semi-finished resulting from the previous stage), the release liner and the backing film. Obtaining the resulting semi-finished product: one or more laminate rolls.
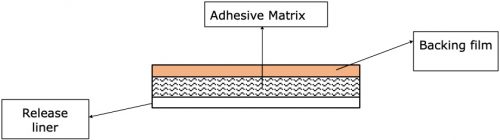
At this stage, it is important to control the weight of the adhesive matrix dispensed per unit area and the adhesive properties of the matrix. In this way, a correct adhesion of the patch at the place of application and a correct dosage of the drug is guaranteed, and consequently the safety, efficiency and quality of the drug.

Die-cutting and packaging
The next and last process is the die- cutting and packaging of the transdermal patches. In this stage, the shape of the patch itself is obtained with the surface established according to the dose. This process is carried out by die cutting the layers obtained in the previous process where several cuts are generally made, one of them being the cut for easy detachment, therefore allowing the patient to open the patch and then to apply it on the safer and more practical area of the skin (as it was explained on this article)
The raw materials that feed this process, in chronological order of their use, are the following: the laminate obtained in the previous stage, release liner, foil for making envelopes and other necessary packaging material.
In this process, the appearance of the patches is controlled to ensure that the cuts are made correctly, as well as any other aesthetic abnormality of the patch that puts the quality of the units produced at risk. Adittionally, a quality control is carried out on the heat-sealed envelopes, thus guaranteeing the hermetic protection of the patches, ensuring their quality throughout their useful life.

Conclusion
As we reviewed, the 3 stages of transdermal patch production require specific adjustments associated with experience, which determines that this is one of the most artisanal pharmaceutical forms available in the industry. This is why, if you or your company are considering working on a transdermal patch development and manufacturing project, it is very important to work with a professional and experienced partner who can demonstrate sufficient knowledge to ensure the necessary quality of this production.
Subscribe to
#AmarinNews
Partner with us!
We offer expertise and experience, together with flexibility and the ability to adapt to your needs.
- info@amarintech.com.ar
- +54 11 4588-6500
- Sanchez 2045 (C1416BQG), Buenos Aires, Argentina.

