Topical and Transdermal Drug Products:
Making the difference through transdermal delivery systems
By Carla Scaramella – Commercial Assistant
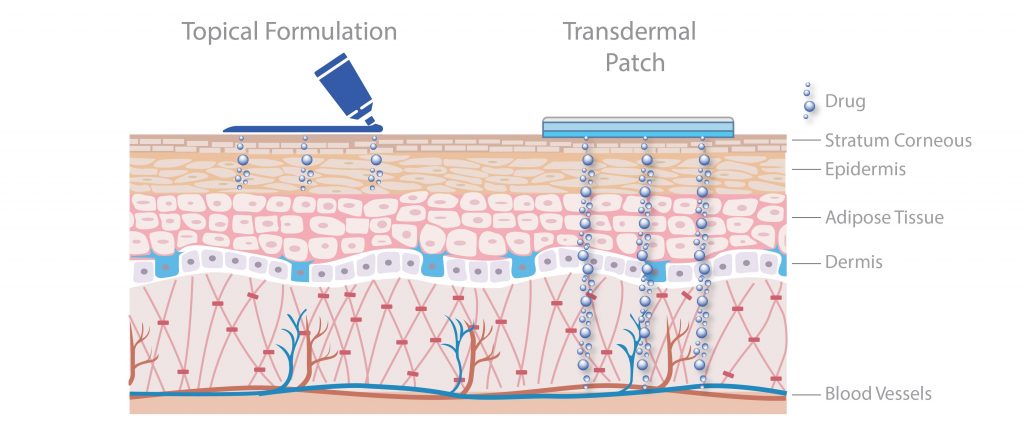
The terms topical and transdermal are sometimes confused. The fact that, by definition, all medications applied onto the skin are topical -that is, applied to the skin surface- may lead to such misconception. However, the term topical product involves the application of a drug on the skin to produce local effects at the site of application, avoiding systemic effects. Therefore, it is designed to minimally penetrate the skin layer.
Topical products are presented in many pharmaceutical forms. The most commonly used are the semisolid dosage forms, which include creams, gels, plasters, ointments and lotions.
Alternatively, transdermal products refer to pharmaceutical compounds that are applied on the skin, but cross the outermost layer of it -the skin barrier- for targeting an effect on more distant tissues or organs.
Such products require a special technology to control drug release and permeation through the skin barrier, in order to achieve a sustained dosage into the systemic circulation. According to the therapeutic needs and drugs’ properties, the delivery duration of a Therapeutic Transdermal System may range between 12 hours and 7 days.
Which are the advantages of a transdermal patch?
Enhances patient compliance with treatment in several ways:
- Non-invasive, pain free, self application, improving the administration of well-known molecules.
- Avoidance of frequent dosing administration, providing a controlled and sustained drug release for defined time periods -involving a lower peak plasma concentration compared to oral forms-.
- Reduction of side effects through avoiding both the gastrointestinal tract -minimizing local irritative GI effects- and the first-pass metabolism -safer in hepato-compromised patients-.
- Versatility for special patient populationts, as it can be administered to a patient who is sleeping, unresponsive, or unable to swallow oral medications.
How are transdermal patches used?
The body location for the patch application may vary with the therapeutic category of each drug, such as chest, buttocks or abdomen, upper torso or upper outer arm. The site of application should be clean, not oily, hairless; and the skin should be intact (unbroken).

What about safety?
Application site reactions may include irritation, erythema, and pruritus. Obviously, other adverse reactions and interactions will depend on each drug safety profile. Patches are safer than oral or injected products, as -unlike them- once an adverse reaction to the Transdermal System is noticed, the patch can be removed and drug administration stops.
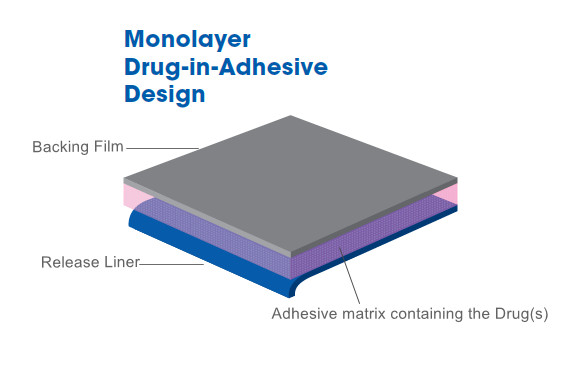
Which are the typical components of the transdermal patches?
-A backing layer (barrier) to protect the patch from the outside environment, including water
-A drug reservoir matrix with or without a semipermeable membrane to control the release of the drug
-A contact adhesive to glue onto skin
-A protective liner to preserve the patch and adhesive, which is removed before applying it.
There are currently 23 transdermal patches approved by FDA. See in Table 1 the chronological list of the most remarkable drugs composing them.
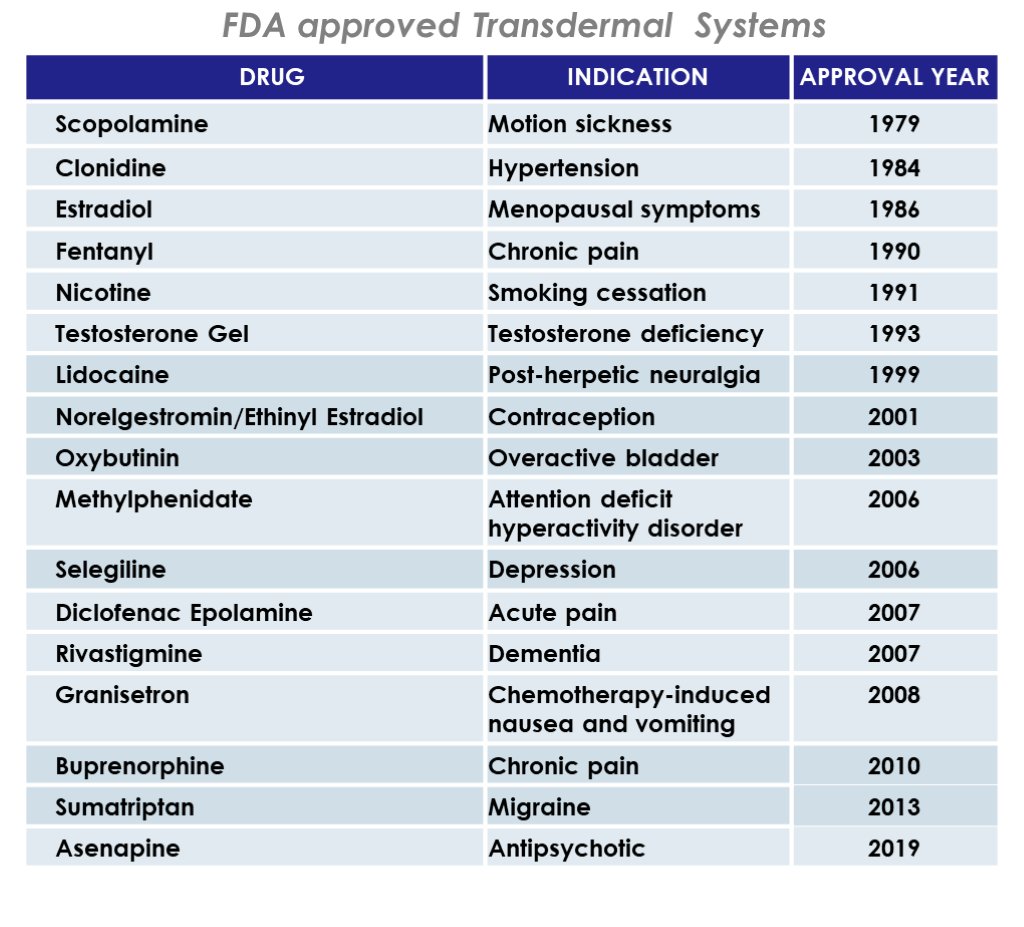
This small number of marketed products reveals the fact that transdermal delivery is limited in terms of the drugs that can be formulated as a transdermal patch. These facts prompt the transdermal companies to be experts in all the aspects of this niche field, involving product formulation, development, clinical trials and manufacturing.
The good news is that, thanks to the development of new technologies, such as novel adhesives, nanoparticles and chemical permeation enhancers to improve drug solubility, the available options for transdermal drug delivery are expanding.
References:
FYs 2013 – 2017 GDUFA Science and Research Report: Transdermal Drug
Products – 23-Apr-2018
2. Transdermal drug delivery. Prausnitz, M., Langer, R. Nat Biotechnol 26, 1261–1268 (2008).
https://pubmed.ncbi.nlm.nih.gov/18997767/
3. Topical and Transdermal Drug Products. Pharmacopeial Forum Vol. 35(3) [May–June 2009]
https://www.uspnf.com/sites/default/files/usp_pdf/EN/USPNF/transdermalStimArticle.pdf
Subscribe to
#AmarinNews
Partner with us!
We offer expertise and experience, together with flexibility and the ability to adapt to your needs.
- info@amarintech.com.ar
- +54 11 4588-6500
- Sanchez 2045 (C1416BQG), Buenos Aires, Argentina.

