Drug-In Adhesive (DIA) patches, a simple design surrounded of demanding challenges
The history began…and followed
Far away, in 1979, the first transdermal patch to reach the market, produced under an industrial controlled process and backed by clinical studies, was launched in US under the brand name TransderScop®. The actually novel product, was developed and produced by ALZA Corporation after years of a research and thanks to a creative and multidisciplinary team. Novartis Consumer Health was not only the sponsor and the marketing holder, but also who made a new pharmaceutical dosage form successfully get into the prescription market. More than 40 year later, the product is still available to consumers for the prevention of motion sickness at sea.
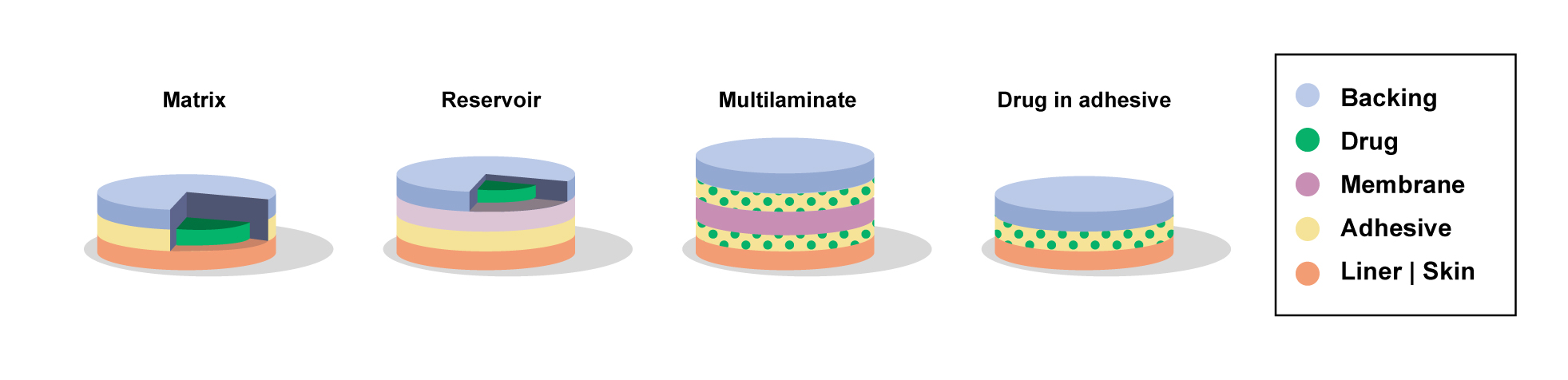
The product is configurated as a multilayer patch composed by a backing film, a drug reservoir holding scopolamine, a drug flux controlling membrane, a contact adhesive and disposable film which is peeled away prior to patch is applied behind the ear. The device is as smaller as a 2.5 cm2 circle programed to deliver 1.5 mg of scopolamine over 3 days.
A relevant milestone was achieved, fruit of scientific and technical efforts although surely accompanied of undesirable drawback and encouraging forwards.
Subsequentially, taking advantage of this emerging technology for drug administration through the skin, three nitroglycerin transdermal patches of different designs were introduced onto the US market in 1981 for the prevention and treatment of angina pectoris: Transderm-Nitro® (Ciba Pharmaceuticals Company) a liquid reservoir patch, Nitro-Dur®, a drug in adhesive patch (Key Pharmaceuticals) and Nitrodisc® a non-adhesive matrix patch with annular adhesive (Searle Laboratories)
What follows is an avalanche of products launched for various indications containing drugs that are released from patches of different designs, reaching 20 different drugs administered through this dosage form. Considering copycat, non copycat and authorized generics there are far more than 100 products available in markets around the world.
US Launch | Drug | Brand name | Patch design |
1979 | Scopolamine | Transderm-Scope | DIA + membrane |
1981 | Nitrogliceryn | Transderm-Nitro | Liquid Reservoir |
1984 | Clonidine | Catapres-TTS | Liquid Reservoir |
1986 | Estradiol | Estraderm | Liquid Reservoir |
1990 | Fentanyl | Duragesic | Liquid Reservoir |
1991 | Nicotine | Nicoderm | DIA |
1993 | Testosterone | Testoderm | Matrix |
1998 | Estradiol + Norethindrone | Combipatch | Liquid Reservoir |
2001 | Ethinyl estradiol + Norelgestromin | Ortho Evra | DIA |
2003 | Estradiol/Levonorgestrel | Climara Pro | DIA |
2003 | Oxybutynin | Oxytrol | DIA |
2006 | Methylphenidate | Daytrana | DIA |
2006 | Selegiline | Emsam | DIA |
2007 | Rotigotine | Neupro | DIA |
2007 | Rivastigmine | Exelon | DIA |
2008 | Granisetron | Sancuso | DIA |
2010 | Buprenorphine | Butrans | DIA + overlay adhesive |
2019 | Asenapine | Secuado | DIA |
2020 | Levonorgestrel + Ethinyl estradiol | Twirla | DIA + membrane + overlay adhesive |
As can be noted in the table, over the years, there was a substantial trend in favor of Drug-In-Adhesive (DIA) patches to reach the market while reservoir patches appear to slowly disappear from the scene.
Liquid Reservoir Patches Have Written Part of the History
Reservoir patches brightened in the 90`s and were the first products for transdermal administration of Nitroglycerine, Clonidine, Estradiol, Norethindrone and Fentanyl.
They were successful in the controlled administration of drug for product lasting several days, mostly 3 to 4 days. All of them has the same structure, a liquid contained in “bag” made with an occlusive film and a semi-permeable membrane in contact with and adhesive which fixes the patch to the skin. The composition of the solvent, the drug load and the permeability of the membrane work all together to achieve the desirable release rate by a diffusional mechanism. A smart idea that requires a smart production process to create an efficient delivery device.
Owing to a few difficulties found in the performance of this devices, including market recalls of several batches due to leakages of the liquid contained in the reservoir, which provoked uncontrolled administration of high doses of the drug, which were even associated with deaths, Health Authorities have put their gaze on the actual safety of this devices.
Additionally, the easy access to the drug allocated in the liquid reservoir, made reservoir patches an ideal source for drug abuse in particular for devices containing narcotic drugs like Fentanyl.
The exposed above, plus the complex production process required to manufacture reservoir patches, made these devices an “specie in extinction” which, in contraposition, offers a field full of opportunities for Drug-In-Adhesive patches.
What a Drug-In-Adhesive patch is.
It is as simple as a of drug loaded in an adhesive, sandwiched between a backing layer and release liner peeled away just before patch application.
A simple device that can be produced by applying a technology widely spread and legated from others industries like those producing adhesive tapes, labels and decals.
Simple to say, not so simple to accomplish in an all-in-one device
The simplicity of a Drug-In-Adhesive patch is just an apparent view of this complex dosage form
There are a considerable number of attributes to achieve to became a secure, efficient approvable pharmaceutical dosage form.
A Drug-In-Adhesive patch should be able to “house” the drug substance and keep it available for diffusion without significant changes during its entire shelf life. It means that it should not only be chemical stable (avoiding drug degradation and impurity formation) but also physically stable. This last condition refers to avoiding the appearance of crystal growth, for those molecules prone to form ordered structures, or to the exudation of the drug in the case of liquid molecules at room temperature.
The adhesive selected plays a mayor roll. It should offer enough solubility to hold a suitable drug load but, at the same time, should not exert a strong drug entrapment to allow the drug substance to freely diffuse toward the skin. The adhesive should assure an adequate adhesive performance, enough to hold the patch stick on the skin for the desired wearing period. At the same time, it should allow a comfortable detachment of the patch after dosing. Moreover, the adhesive should not provoke irritation and sensibilization of the skin.
Patches designed to last more that 4 days usually requires and additional layer on top of the backing film in order to create a peripheral adhesive surface. They are named patches with overlayed or peripheral adhesive. Such design increase formulation and manufacturing complexity.
Depending on the drug substance and characteristic, the composition formulated might consider the addition of functional and non-functional excipients.
Functional excipients are those who impart a substantial effect on the bioavailability of the drug and the release profile. In transdermal patches they are recognized as permeation enhancers. Many substances have been used for that purpose. Its inclusion in a patch requires a delicate balance since they use to jeopardize the adhering properties of the adhesives.
Non-functional excipients are commonly considered when the adhering properties of the adhesive need to be modified to increase or decrease tackiness, when the drug substance requires physical or chemical stabilization to avoid either crystallization process or drug degradation or both.
So far, we have been talking about the fill of the sandwich. Now we have to talk about the “bread”
A backing film should be able to strongly anchor the adhesive to facilitate the production process and to avoid detachment when the patch is removed from the skin
Occlusion properties of the backing film can influence drug permeation. The selection of materials should take this aspect into account. The backing film should also offer a comfortable use, diminishing incommodities for the patients and are aesthetical acceptable. There are customers who prefer it make the patch as invisible as possible while other are more prone to evidence the patch presence, therefore, color and texture are part of the material selection process.
The release liner is the other slice of the bread. The main function is to accompany the patch, from the manufacturing process itself, and then provide a smooth detachment to facilitate the application of the patch. The material selection deserves attention since such properties should be kept all along the shelf life of the patch with null interaction with API and excipients. There were a few cases of market recalls due to complains raised for not being possible to peel off the release liner from the patch.
We have now the sandwich made; we have to pack it. In most of the cases patches are individually packet in impermeable pouches.
Primary packaging material are in close contact with the patch so extractables and leachables are a matter of concern and should be considered during material selection. However, this is a consideration for any dosage form. In patches, either the drug substances or some excipients can be volatile at room temperature and can easily migrate and diffuse to the inner liner of the foils need to pouch the patches. In these cases, the selection of materials with barrier properties to the migration of APIs and functional excipients must be taken into account.
Conclusion,
Adhesive Drug Patches are complex dosage forms, based on a simple industrial design. Although it is a great challenge for development and manufacturing, the current availability of excipients specifically designed for this dosage form and supplied by well-recognized suppliers in the field, greatly facilitates the formulation development process.
Specific and key machinery is required to obtain a robust and sustainable process. Many machine manufacturers in the adhesive application and film converting industry have been evolving and adapting their technologies and designs to meet the demand of the patch manufacturer to successfully feed the growing market with quality products that this pharmaceutical form represents.
After more than 30 years on the niche, Amarin technologies has the capability for both, formulate Drug-In Adhesive patches and manufacture it on demand.
Do not miss us to contact us for a patch design.
Subscribe to
#AmarinNews
Partner with us!
We offer expertise and experience, together with flexibility and the ability to adapt to your needs.
- info@amarintech.com.ar
- +54 11 4588-6500
- Sanchez 2045 (C1416BQG), Buenos Aires, Argentina.

